Heat Capacity
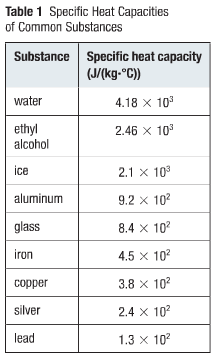
The amount of energy needed to raise the temperature of 1kg substance by 1○C is
called the specific heat capacity. This amount of energy is
measured in joules, every substance will have a certain specific heat capactiy,
as we can see in the table on the left. Quantity of heat (Q) is the amount of thermal energy transferred from one object to another. A calculation can be made (in Joules) to calculate the quantity of heat, where m is the mass, c is the specific heat capactiy and (∆T) is the change in temperature that the substance undergoes:
Q= mc∆T or Q=mc (T2-T1)
called the specific heat capacity. This amount of energy is
measured in joules, every substance will have a certain specific heat capactiy,
as we can see in the table on the left. Quantity of heat (Q) is the amount of thermal energy transferred from one object to another. A calculation can be made (in Joules) to calculate the quantity of heat, where m is the mass, c is the specific heat capactiy and (∆T) is the change in temperature that the substance undergoes:
Q= mc∆T or Q=mc (T2-T1)
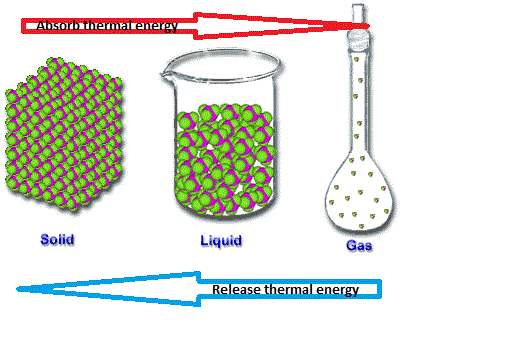
Latent Heat
When solids, liquids or gases change state it is because they absorb or release enough thermal energy. When a solid changes into a liquid, this is known as fusion. When a liquid changes state into a gas, this is called vapourization.
Heating and Cooling Graphs
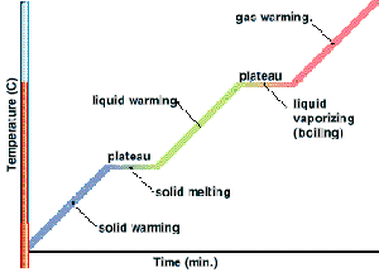
Example of a heating graph
We can graph the changes in temperatures using heating and cooling graphs. A heating graph will show the temperature changes when a substance absorbs thermal energy (heats up). Likewise, a cooling graph shows the temperature changes of a substance that loses (releases) thermal energy (cools down). In both graphs, the y-axis is the temperature in Celsius, and the x-axis is the amount of thermal energy released or absorbed. The angled parts of each graph show the temperature change and occur when only one state is present. The flat parts of the graph occur when there are more than one state present, and show a constant temperature because one state is changing into another. A heating graph will show a clear boiling point and melting point, whereas a cooling graph will show the condensation and freezing point.
Latent Heat
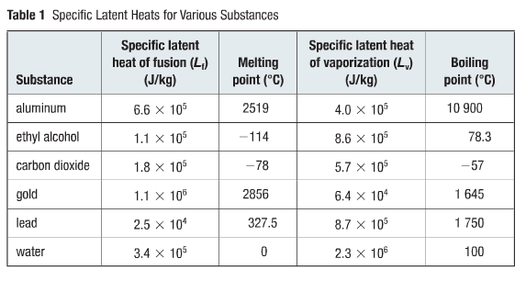
The released and absorbed thermal energy during a change of state is the latent heat (Q) of the substance. Every substance has a latent heat of fusion and vapourization. The latent heat of fusion will be the amount of thermal energy required to change a solid into a liquid, or vice versa. The latent heat of vaporization is the amount of thermal energy required to change a liquid into a gas, and vice versa. Specific latent heat (L) of a substance is the amount of thermal energy required for 1kg of a substance to change from one state into another. So the specific latent heat of fusion (Lf) is the thermal energy needed to cause 1kg of a substance to melt or freeze. Lastly, the specfic latent heat of vapourization (Lv) is the thermal energy required for 1kg of the substance to boil or condense. We can use the table (Table 1- above) and the equations below to calculate specific latent heat.
Practice
Simulations
1. http://phet.colorado.edu/en/simulation/states-of-matter (changes of state)
2. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=655&ClassID=2057186 (heat absorption)
Quizzes/Worksheets
1. http://www.kearsarge.k12.nh.us/1112208220251417/lib/1112208220251417/pb_10d.pdf (worksheet)
2. http://www.sciencegeek.net/Chemistry/taters/Unit7Thermochemical.htm (fill in blank)
3. http://www.algebralab.org/practice/practice.aspx?file=Algebra_SpecificHeatCapacity.xml (specific heat capactiy practice problems)
1. http://phet.colorado.edu/en/simulation/states-of-matter (changes of state)
2. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=655&ClassID=2057186 (heat absorption)
Quizzes/Worksheets
1. http://www.kearsarge.k12.nh.us/1112208220251417/lib/1112208220251417/pb_10d.pdf (worksheet)
2. http://www.sciencegeek.net/Chemistry/taters/Unit7Thermochemical.htm (fill in blank)
3. http://www.algebralab.org/practice/practice.aspx?file=Algebra_SpecificHeatCapacity.xml (specific heat capactiy practice problems)