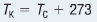Kinetic Molecular Theory

Heat, energy and temperature are everywhere in our everday lives. The kinetic molecular theory helps to explain warmth and coldness. It is the theory that describes the motion of molecules or atoms in a substance in terms of kinetic energy. It is based on the idea that matter is composed of particles that attract each other and have kinetic energy. This kinetic energy is what causes atoms or molecule to be in constant motion, the forces of attraction between molecules varies on the state of matter. For example, as seen below, the forces of attraction in a solid are in a fixed position and vibrate because of the kinetic energy they posses. When the solid begins to warm up, the particles will stay in the fixed positions, but will vibrate more rapidly. So this increase in kinetic energy will make the substance feel warmer, and vice versa for a decrease in kinetic motion. The particles of a gas and liquid vibrate more rapidly than a solid and move around.
Thermal Energy and Temperature
Thermal energy is the total amount of kinetic and potential energy possessed by the atoms or molecules of a substance. The kinetic energy which is associated by the motion of particles, and potential energy is associated with the atom bonds. Objects warm up when absorbing thermal energy and cool down when releasing it. Thermal energy can only be transferred to warmer to colder objects. Measuring the average kinetic energy of particles can give an indication how hot or cold an object is. Temperature is the meausure of the average kinetic energy of the particles in a substance. There are various scales used to measure temperature, for example, Celsius scale, Farenheit scale and the Kelvin scale. To convert from the Kelvin scale to Celsius scale (and vice versa) we can use this equation:
Heat
We know that the thermal energy determines an object's temperature, so two identical object at the the same temperature will have the same amount of thermal energy, but this is only true if the objects are made of the same material and have the same mass. The difference between thermal energy and heat is that heat, is the transfer of thermal energy from a warmer object to a colder one (higher temperature to lower temperature). There are many ways that thermal energy can be transferred, the three main methods are thermal conduction, convection and radiation. Thermal conduction is the transfer of thermal energy that occurs when warmer objects are in physical contact with colder objects. Usually occuring in solids, when heat is applied to on end of an object, the particles at that end will begin to move more rapidly. This causes the molecules to bump into one another and transfer kinetic energy.

Convetion occurs when the particles of a lot of heat energy in a liquid or gas move and take the place of particles with less heat energy. The colder denser fluid will fall and this then pushes up the warmer, less dense fluid. When this repeats, it is called a convetion current, which occurs when a fluid is continously heated; caused by warmer, less dense fluid being pushed up as colder, denser fluid falls downard.
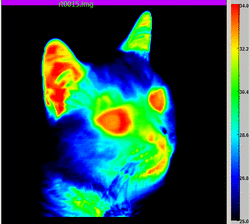
Radiation is simply the movement of thermal energy as elctromagnetic waves. This
can come from sources such as the Sun, flames and lamps. In the picture on the left, a thermal imaging camera detected infrared radiation and produced an image called a thermogram.
can come from sources such as the Sun, flames and lamps. In the picture on the left, a thermal imaging camera detected infrared radiation and produced an image called a thermogram.
Practice
Simulations/Videos
1. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=658&ClassID=2057186 (convection and conduction)
2. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=388&ClassID=2057186 (conduction)
3. http://www.bbc.co.uk/schools/gcsebitesize/science/aqa/heatingandcooling/heatingact.shtml (video)
Quizzes/Worksheets
1. http://www.kearsarge.k12.nh.us/1112208220251417/lib/1112208220251417/PB_10A.pdf (converting Celsius to Kelvins)
2. http://www.bbc.co.uk/apps/ifl/schools/gcsebitesize/science/quizengine?quiz=aqa_heatingtest&template (multiple choice- basic concepts)
3. http://www.physicslessons.com/quiz/quiz10.html (thermal energy- multiple choice quiz)
1. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=658&ClassID=2057186 (convection and conduction)
2. http://www.explorelearning.com/index.cfm?method=cResource.dspDetail&ResourceID=388&ClassID=2057186 (conduction)
3. http://www.bbc.co.uk/schools/gcsebitesize/science/aqa/heatingandcooling/heatingact.shtml (video)
Quizzes/Worksheets
1. http://www.kearsarge.k12.nh.us/1112208220251417/lib/1112208220251417/PB_10A.pdf (converting Celsius to Kelvins)
2. http://www.bbc.co.uk/apps/ifl/schools/gcsebitesize/science/quizengine?quiz=aqa_heatingtest&template (multiple choice- basic concepts)
3. http://www.physicslessons.com/quiz/quiz10.html (thermal energy- multiple choice quiz)